Insights about sensing mechanisms with Nickel oxide based gas sensors
Project Director: Dr. Cristian Eugen SIMION
Among different types of sensors, those based on semiconducting metal oxides stands out through their: robustness, selective sensitivity, low fabrication costs and infield operation (presence of the relative humidity, variable temperature, the presence of potential interfering gases, etc.).
The idea of the current project proposal consists in exploring the intrinsic nature of Nickel Oxide (NiO) as sensitive material together upon inspired chemical synthesis pathways, merging towards extracting the insights of its gas sensing performances towards different target gases (CO, CO2, NO2, CH4, NH3, SO2, H2S) adapted to work under infield conditions.
The research work employs interdisciplinary vision of the team spanning from: chemical-physics and theoretical physics.
The novelties brought by the project are:
- Structure-functioning relationships will be highlighted by understanding the role of sensing and transducing features with multi-dimensional NiO nanostructured based sensors. Through inspired chemistry synthesis, NiO morphologies will be tailored to maximize the gas sensing performances.
- The challenge is to fuse the theoretical predictions with the realistic gas sensing outputs into an overview image about the nature of gas surface interactions.
The outputs from phenomenological and catalytic investigations will guide the theoretical modeling towards extending the gas surface model of NiO sensors.
1. NiO materials with controlled morphology.
2. Complete electronic library with NiO network parameters, elemental composition, crystallite size distribution, volume and pore size distribution.
3. Different batches of NiO-based sensors accompanied by fully documented procedures.
4. Selection of gas-sensitive layers without surface cracks and agglomerations as a result of optical inspection.
5. Selection of sensors following the evaluation of the parameters of: sensitivity, selectivity, response time and return time as well as electricity consumption.
6. Selection and separation of NiO-based materials according to the nature of the layers (completely or partially enriched in majority charge carriers) in accordance with the theoretical analysis of gas-sensitive properties.
7. Distinction between physisorption and ionosorption processes, individually represented for NiO sensors partially or completely enriched in majority load carriers.
8. Quantification of surface reactions using the formalism of quasi-chemical equations
9. Modeling the effects associated with load exchange processes by using the concepts of solid state physics (for example, load loading statistics, theory of transport phenomena, etc.)
| Cristian Eugen Simion |
| Adelina Stanoiu |
| Cosmin Istrate |
| Catalina Mihalcea |
| Ovidiu Gabriel Florea |
| Corneliu Ghica |
| Andrei Cristian Kuncser |
| Ion Viorel Dinu |
| Daniela Ghica |
| Ionel Florinel Mercioniu |
| Simona Somacescu |
Summary of the activities carried out during: 01.01.2021 – 31.12.2023
During the implementation period of the project: Perspectives on detection mechanisms with gas sensors based on nickel oxide PCE 116 / 2021 ( PN-III-P4-ID-PCE-2020-0506) the following activities were carried out:
2021 Stage 1 - Materials gas - sensitive based on NiO with structure and morphological specific properties
Act 1.1 - Preparation of gas-sensitive materials based on Nickel oxides (NiO).
Four types of Ni oxide based powders were prepared using different chemical routes by hydrothermal synthesis assisted by different structural directing agents.
Act 1.2 - Morphological and structural properties of NiO-based materials.
It was observed that the chemical synthesis route directly influences the morpho-structural functions of the nickel oxides both from the point of view of particle size and from the morphological point of view.
Act 1.3 - Fabrication of sensors based on NiO.
NiO-based powders were mixed with an organic solvent (eg α-Terpineol 90%), plastered, deposited on commercial Al2 O3 supports and thermally treated in order to obtain compact sensitive structures prepared for the next stages of the project.
The stage of achieving the 2021 achievement indicators
Two papers published in Chemosensors-MDPI, an ISI listed journal; an Editorial published in Materials-MDPI; a participation in the " Contemporary Solutions for Advanced Materials with high Impact on Society" Workshop, October 11-15, 2021, Bucharest; web page
2022 Stage 2 – Sample selection based on NiO for carrying subsequent activities. Feedback to preparation strategies.
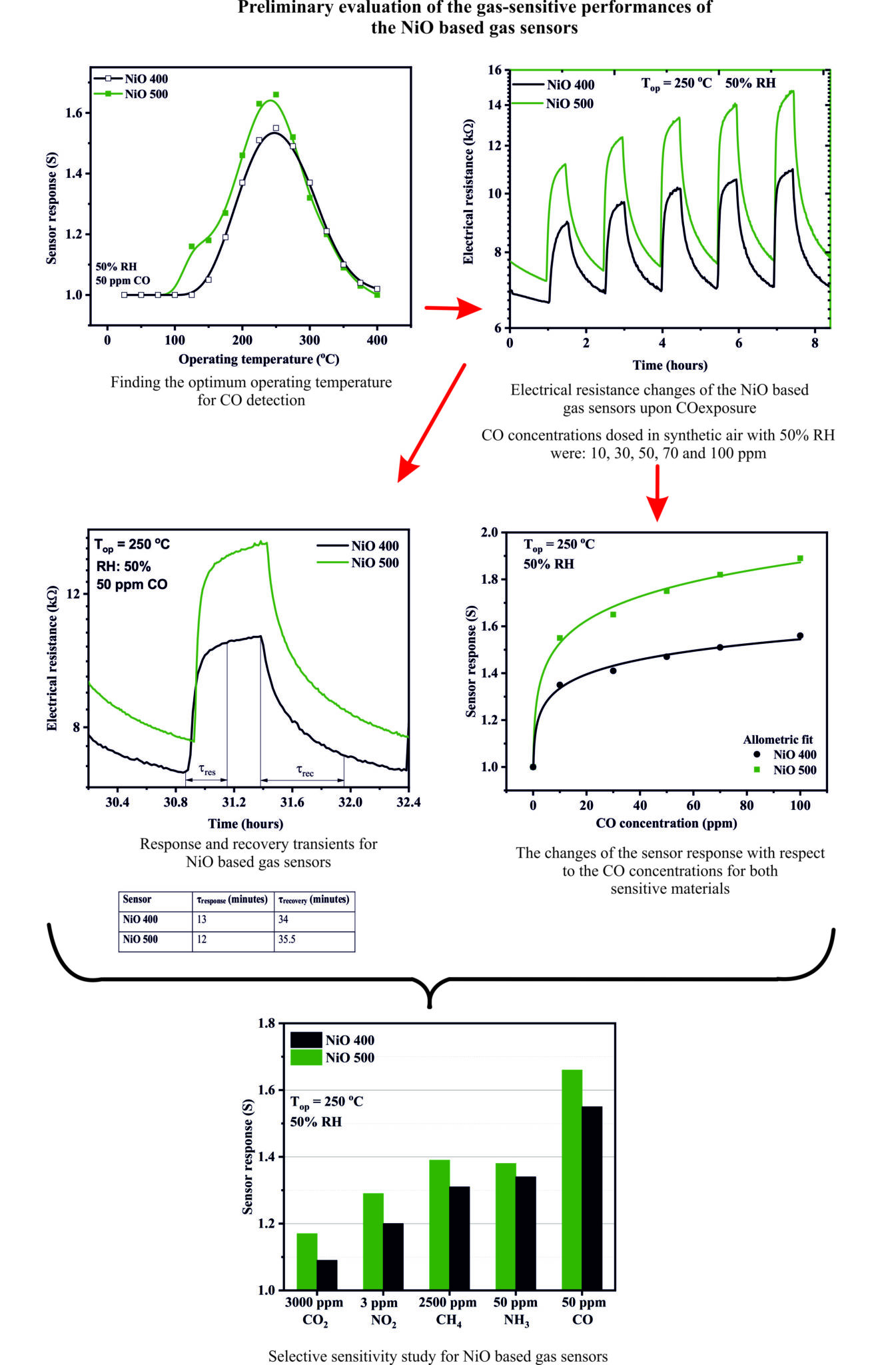
Act 2.1 - Preliminary evaluation of the gas-sensitive performances of NiO-based materials to the following test gases: CO, CO2, CH4, NO2, NH3, SO2, H2S in an atmosphere with controlled humidity (0- 80% RH).
All sensors prepared in Act 1.3 were subjected to preliminary gas-sensing investigations. Among them, the material prepared with Graphene content was excluded due to its high conductivity (eg low electrical resistance under test conditions) as well as the lack of response to CO used as the target gas. In order to understand the role of the calcination temperature on the gas-sensitive performances, the sensors prepared by the same chemical synthesis were chosen where only the calcination temperature was varied (eg NiO 400 – 400 °C respectively NiO 500 – 500 °C)
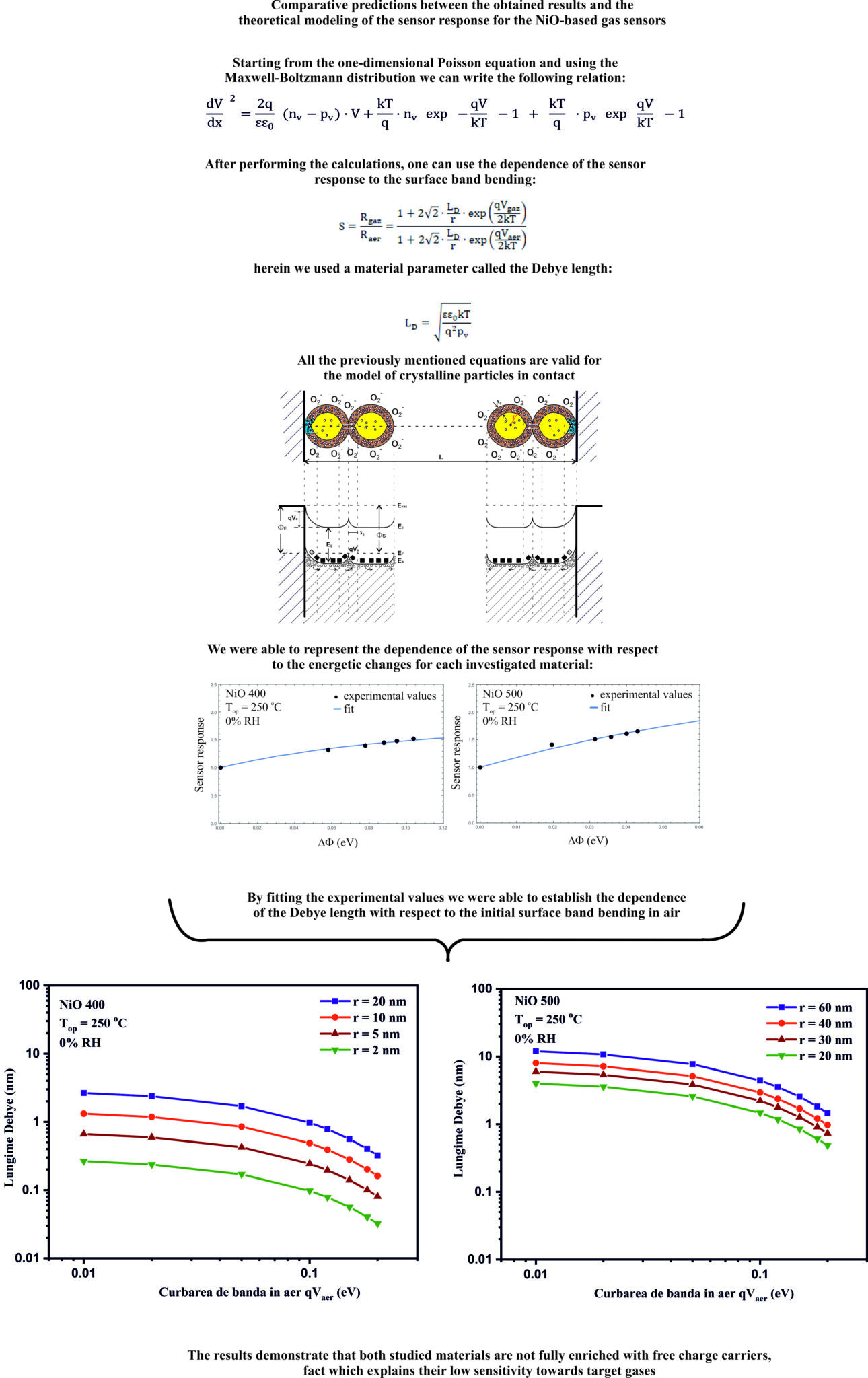
Act 2.2 - Comparative predictions between the obtained results and the theoretical modeling of the sensor response.
The calibration curves for the tested sensors (eg sensor signal as a function of CO concentration) indicated an allometric response, common to materials based on semiconducting metal oxides. Fitting the experimental values allowed us to evaluate the Debye length for each material studied separately, depending on the initial band curvature in air (qV air ) , demonstrating that both studied materials (eg NiO 400 and NiO 500) are not found in the situation of the completely enriched in free charge carriers (eg holes – h + ).
The stage of achieving the 2022 achievement indicators
Two papers published in Chemosensors-MDPI – IF = 4.229, on the subject of the project; a paper published in Sensors and Actuators B. Chemical – IF = 9.221 and one in Materials Letters – IF = 3.574 with thanks to the current project; a participation in the International Conference on Semiconductors (CAS) with an oral presentation during October 12-14, 2022. IEEE Xplore Proceedings 2022 pp. 109-112, https://doi.org/10.1109/CAS56377.2022.9934703; web page.
2023 Stage 3 Realistic modeling of gas-surface interactions through complex investigations.
Act 3.1 – Phenomenological and catalytic investigations.
The unitary approach of the quasi-chemical formalism (surface chemical processes) and the electronic formalism (electronic exchange between the solid and the surface), allows the elaboration of the chemical-physical mechanism underlying the operation of the sensors. From an experimental point of view, this objective involves complex phenomenological investigations, namely simultaneous measurements of electrical conductivity, extraction work and additional catalytic conversion. If the first two allow the evaluation of the transducer function of MOX, the third technique allows the evaluation of the receptor function .
Concretely, the use of the catalytic conversion technique leads to a deep understanding of the involved chemical component (reaction rate, reaction order, influence of the operating temperature of the material) which will be modeled with the help of the formalism of quasi-chemical equations using an adapted notation of the type Kroeger-Vink. The evaluation of the extraction work and the electrical resistance will allow obtaining information about the electrical nature of the reaction species, with or without charge exchange with the gas-sensitive material, which leads to the understanding of surface interactions and the development of possible interaction mechanisms. The advantage of complex phenomenological investigations carried out with the Dynamic Gas Mixing System (SMG) is the precise reproduction and long-term maintenance of a calibrated concentration of test gas (in this case carbon monoxide) dosed with high precision in the ppm range. From an application point of view, the proposed phenomenological approach requires the introduction of a key element permanently present in the operating conditions in the field, namely the existence of atmospheric oxygen and relative humidity (RH %).
The expected results based on the techniques involved can be summarized as follows:
Catalytic conversion (ΔX) – the variation in the amount of water vapor (in % @ constant temperature or ppm) before and after sensor exposure as well as the amount of carbon dioxide (CO 2 ) resulting from the catalytic process of burning carbon monoxide.
Electrical resistance (ΔR) – the variation in the concentration of free charge carriers before ( R_i)and after (R_f) exposure to CO under normal operating conditions (eg the presence of RH in different percentages, constant pressure, constant gas flow).
The work function (ΔΦ) – simultaneously with the monitoring of ΔR, allows to explain the contribution brought by the variation of electronic affinities (Δχ). The importance of the quantitative assessment (energy) of the electronic affinity lies in the fact that it brings information about the dipolar species in the form of hydroxyl groups linked to the metal atom that do not involve charge transfer (ΔR=const.) but prevent or favor subsequent interactions with the test gases .
Thus, through the unitary approach of the quasi-chemical formalism together with the electronic formalism, the realistic chemical-physical mechanism involved in the sensing process resulting from the involvement of oxygen and water vapor can be elaborated.
The material marked with NiO1 400 shows a typical p- type semiconductor behavior of the sensitive material (NiO). Such a phenomenon can be explained by the reducing effect of carbon monoxide on the pre-adsorbed surface oxygen species, releasing electrons in the valence band, thus decreasing the total concentration of holes (as majority charge carriers). Moreover, the negative charge carriers captured on the surface (due to the pre-adsorbed oxygen species) are completely electrically balanced by the gaps located in the near region in the accumulation layer, satisfying the electroneutrality condition. To determine how electrical conduction controls the displacement of the Fermi level (Debye length > crystallite radius), or the change in the height of the potential barrier (Debye length < crystallite radius) for the case of NiO we refer to the relationship below. Regarding the quantitative analysis (∆Φ; q∆Vs and ∆χ) this provides clear evidence that CO exposure induces a slightly pronounced decrease in both ∆Φ and aq∆Vs in the presence of RH, while the electron affinity ∆χ remains almost unchanged. Such a particular aspect amplifies the idea of the reaction yield between CO with either pre-adsorbed oxygen species or network oxygen rather than direct interaction with physisorbed water.
In the case of NiO1 500 It is observed that both the electrical resistance (R) and the contact potential difference (CPD) increase with increasing CO concentration, when the sensor is operated at a temperature of 250°C in conditions of 50% relative humidity . It is observed how with the increase of carbon monoxide concentration, both the work function and the curvature of the energy bands on the surface decrease monotonically.
On the other hand, the electron affinity increases for the first exposure to CO (respectively 15 ppm) followed by a capping of its variation. Consequently, a mechanism with dual involvement in CO sensing can be proposed.
The increase in electron affinity can be explained by considering the dissociation of water at the gas-sensitive surface. Consequently, an interaction between CO and surface hydroxyl groups is likely to occur with the release of electrons into the bulk of the material (eg, leading to a decrease in surface band bending).
On the other hand, the electron affinity remains constant above 15 ppm CO. A possible surface interaction mechanism is proposed and can be separated into two processes.
The first, as I said above, consists in the interaction of CO with the hydroxyl groups on the surface, and the formation of new OH bonds with the release of electrons in the NiO volume, and the second consists in the direct interaction between CO and oxygen pre-adsorbed with formation of CO2 as a reaction product and of course with the release of electrons that contribute to the recombination with the holes in the surface accumulation layer.
For the gas-sensitive material NiO2 400, the electrical and energetic behavior was followed when exposed to carbon monoxide in the range (15-100 ppm). The previously determined optimum operating temperature was set and kept constant throughout the investigations at 250°C.
It was observed that while the extraction work and surface band bending decrease slightly over the entire range of CO concentrations, the electron affinity is constant within measurement errors. Such behavior can be explained by associating the interaction between CO only with the pre-adsorbed oxygen species and not with the OH groups on the surface.
NiO 500 shows the highest sensor signal in the presence of relative humidity.
This fact is remarkable because if we look at the associated catalytic conversion graph, we notice the lack of catalytic conversion from CO to CO2 under 50% RH conditions. This indicates either the reuse of CO2 in the subsequent sensing processes corroborated with the lack of catalytic conversion on the interdigital structure of Pt electrodes.
The two materials NiO1 400 and NiO2 400 show similar behavior, which is also found in the values of the sensor signals. Catalytic behavior is found in both sensors and can be translated by a strong carbon monoxide combustion effect followed by its conversion to CO2 (both in 0% RH and 50% RH atmospheres). We can conclude that the effects are approximately linearly dependent as previously described due to the main combustion on the sensitive layer followed by the conversion on the Pt interdigital electrodes.
Act 3.2 – Complete modeling of gas-sensitive surface interactions for NiO-based materials – the link to reality.
In the case of NiO type gas-sensitive materials, the effects of exposure to carbon monoxide on the extraction work, indicated very pronounced charge transfer processes which, however, are not reflected in the electrical resistance changes of the sensitive layer. This experimental finding should be viewed carefully given the fact that p -type conducting oxide materials are much less used as gas-sensitive materials for chemo-resistive gas sensors than n -type conducting oxide materials . A first explanation was proposed taking into account the fact that the adsorption of oxygen causes the appearance of an accumulation layer on the surface of the nickel oxide, and if we consider the equivalent circuit of the sensitive layer as a parallel circuit of resistances corresponding to the accumulation layer on the surface of the crystallites (small as value) and volume (large), we can see that the resistances that change, those of the surface layer, are the small ones and that the impact in the total resistance of the layer will be reduced. In the case of materials with n- type conduction , we encounter exactly the opposite situation, because the resistances corresponding to the surface layer, in their case, the depletion layer, are much higher than the resistances unaffected by the surface effects with which they are lined.
The stage of achieving the achievement indicators 2023
published work in the Sensors and Actuators B. Chemical, a paper under review in the Applied Surface Science Ms. Ref. No.: APSUSC-D-23-15783 Title : Atomic-level insight into the structural properties of nanoparticles NiO for CO sensing on the topic the project ; an accepted paper in the Journal Physica E: Low-dimensional Systems and Nanostructures with thanks to the current project ; a participation in the Conference International of Semiconductors (CAS) with presentation invited in the period 11-13 October 2023 IEEE Xplore Proceedings 2023 pp. 11-18, https:// doi.org/10.1109/CAS59036.2023.10303715 ; a participation in the Conference International of Semiconductors (CAS) with presentation oral in the period 11-13 October 2023 IEEE Xplore Proceedings 2023 pp. 63-66, https:// doi.org/10.1109/CAS59036.2023.10303672 web page .
2021 Stage 1 - Materials gas - sensitive based on NiO with structure and morphological specific properties
Complete chemical synthesis route, can be found here:

Complete XRD report can be found here:
XRD pattern and data library:
| Material | Calcination temperature | Crystalline phase | Lattice parameter | Particle size |
| NiO-1 | 400°C | Cubic Fm-3m (225) | a = 0.4178 ± 0.0001 nm | d = 11 ± 2 nm |
| NiO-1 | 500°C | Cubic Fm-3m (225) | a = 0.4178 ± 0.0001 nm | d = 18 ± 2 nm |
| NiO-2 | 400°C | Cubic Fm-3m (225) | a = 0.4178 ± 0.0001 nm | d = 18 ± 2 nm |
| NiO-rGO | 400°C | Cubic Fm-3m (225) Hexagonal C-2H (grafit 2H) P63/mmc | aNiO = 0.4177 ± 0.0001 nm agrafit = 0.2460 ± 0.0001 nm și cgrafit = 0.6707 ± 0.0001 nm
| dNiO = 7 ± 2 nm dgrafit = 35 ± 4 nm |
SEM investigations:

TEM and HRTEM investigations:

Specific surface BET (Brunauer, Emmett, Teller) data:
| Material | Calcination temperature | Pore volume (cm3/g) | Pore size (nm) | Specific surface (m2/g) |
| NiO-1 | 400°C | 0.179 | 38.88 | 18.473 |
| NiO-1 | 500°C | 0.162 | 34.12 | 19.075 |
| NiO-2 | 400°C | 0.169 | 33.17 | 20.486 |
| NiO_rGO | 400°C | 0.208 | 38.22 | 21.86 |
Purpose of the procedure: Obtaining batches of four planar sensors based on Nickel oxide, fully prepared for the development of the stages of evaluation of sensitive gas performance. Field of application: Gas sensors based on thick and porous films of semiconductor metal oxides. Nickel oxide powders obtained by different chemical routes are transported and stored in specially designed plastic containers. They must meet the following conditions: - Must not chemically or physically affect the NiO sample; - It must ensure a tight closure so that there is no loss of material, both during transport and when stored; - NiO samples will not be exposed to direct sunlight and will be stored in laboratory conditions.

Weigh 59 mg of each material or 57.5 mg of organic solvent, which is then mixed in a mortar and pestle with minimal roughness and absorption (preferably agate). Grind very well for 10 minutes, so as to obtain a paste with medium viscosity, which is to be transferred to Al2O3 supports, by the following method:
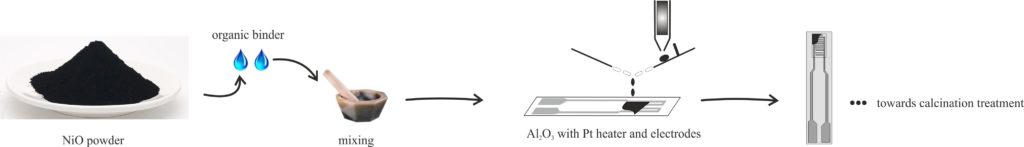
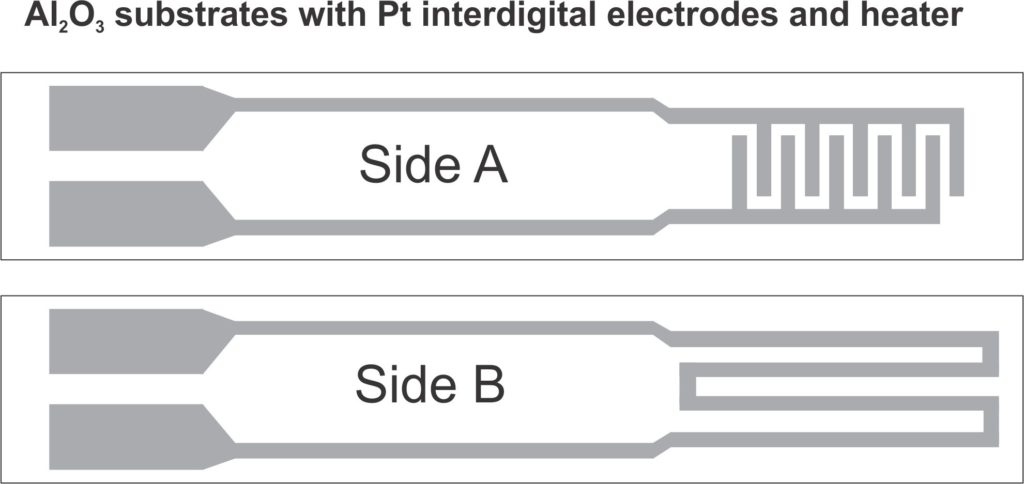
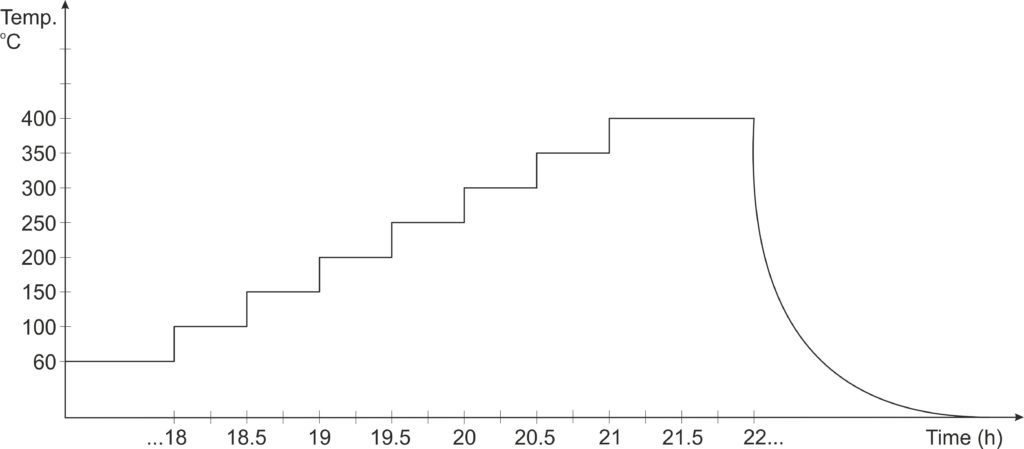
After transferring the paste to the alumina supports, they are placed in the oven at 60 ° C for 18 hours. The final heat treatment consists of the progressive heating of the sensors in a programmable oven, according to the graph below:
In order to determine the operating temperature of the gas-sensitive material, it is needed as an initial a calibration of the Platinum heater takes place, depending on the voltage applied to it, using the electrical configuration below. The optical pyrometer used is of the Lumasense IN-5L Plus type, which is able to detect an infrared emission of the NiO type material, as a result of the heat transfer from the Platinum heater to the gas-sensitive layer. The IR emission was corrected with the NiO emissivity coefficient (ε = 0.95).
The obtained results are in good agreement with those provided in Work Plan of Stage 1.
In conclusion, the objectives and activities proposed for Stage 1/2021 were fully achieved.
Stage 2 – Sample selection based on NiO for carrying subsequent activities. Feedback to preparation strategies.
THE YEAR 2023 Stage 3/2023 Realistic modeling of gas-surface interactions through complex investigations.
ACTIVITY Act 3.1 – Phenomenological and catalytic investigations.
Act 3.2 – Complete modeling of gas-sensitive surface interactions for NiO-based materials – the link to reality. Objectives achieved: - Simultaneous evaluation of extraction work and electrical conductance as well as catalytic conversion. - Interpretation of surface interactions through the prism of quasi-chemical equations. The stage of reaching the achievement indicators - a paper published in Sensors and Actuators B. Chemical, a paper under review in Applied Surface Science Ms. Ref. No.: APSUSC-D-23-15783 Title: Atomic-level insight into the structural properties of nanoparticulate NiO for CO sensing, on the subject of the project; a paper accepted in the journal Physica E: Low-dimensional Systems and Nanostructures with thanks to the current project; a participation in the International Conference on Semiconductors (CAS) with an invited presentation during October 11-13, 2023 IEEE Xplore Proceedings 2023 pp. 11-18, https://doi: 10.1109/CAS59036.2023.10303715; web page.
Deliverable: - Deliverable 3.1.1. Separation of physisorption and ionosorption interactions for each individual case (materials partially or completely enriched in free charge carriers). - Deliverable 3.2.1. Quantification of surface reactions using quasi-chemical equation formalism. - Deliverable 3.2.2. Modeling of charge exchange effects using solid state physics concepts (statistics of charge carriers, theory of transport phenomena). Scientific report 1. Introduction. 2. Complex phenomenological investigations (simultaneous measurements of extraction work and electrical resistance). Details on possible detection mechanisms under field-like conditions. 3. Simultaneous investigations of catalytic conversion and electrical resistance. Details of possible surface interactions. 4. Formalism of quasi-chemical equations. Surface oxygen adsorption and its interaction with carbon monoxide. 5. Possible transport phenomena in NiO-based materials. 6. Selective bibliography. 7. Conclusions of the obtained results. 8. The estimated impact of the results obtained, emphasizing the most significant result obtained. 9. Dissemination.
Complex phenomenological investigations (simultaneous measurements of work function and electrical resistance). Details on possible detection mechanisms under field-like conditions. Nickel oxide (NiO)-based gas-sensitive materials were prepared by different chemical routes and were indexed. Indexed material; Chemical route; Calcination temperature;
NiO1 400 hydrothermal synthesis and ethylene glycol (EG) as structural directing agent 400°C
NiO1 500 hydrothermal synthesis and ethylene glycol (EG) as structural directing agent 500°C
NiO2 400 hydrothermal synthesis and Tripropylamine (TPA) as structural directing agent 400°C
In general, the target gases react with the heated surface at a specific temperature of the semiconductor metal oxide gas sensor and are consequently converted into different products depending on the target gas and the ambient conditions. Under normal air conditions, for example, reducing gases such as CO and H2 are oxidized to reaction products of the form: CO2 and H2O by reaction with pre-adsorbed oxygen species from the surface of the gas-sensitive material (generally considered to be at the origin of the gas sensor response). A powerful and simple method that allows us to get an idea of the reaction pathways and the mechanisms that take place during these reactions is therefore to measure the variations in the composition of the ambient atmosphere caused by the detection process. Basically, the gas composition before and after the sensor chamber is monitored. In our case we used an INNOVA 1314 photoacoustic analyzer equipped with optical filters for CO, CO2, H2O and CH4. For the simplicity of determining possible mechanisms of interaction with the surface of NiO-based sensors, we used carbon monoxide as the target gas in the absence and presence of relative humidity. From the simultaneous measurements of electrical resistance and catalytic conversion we were able to extract the sensor signal in the radar plot representation as well as the concentrations of CO and H2O before the sensor chamber, respectively CO2 and H2O after exposing the heated sensors to 250°C. It should be noted that for better accuracy, two sensors from the same batch, specially built for catalytic measurements, were mounted in the sensor chamber. More specifically, the Al2O3 substrate was covered with glass on the entire surface of the Pt heater, and respectively all the Pt contacts, leaving free space only in the deposition area of gas-sensitive NiO-based materials to reduce the catalytically active surface.
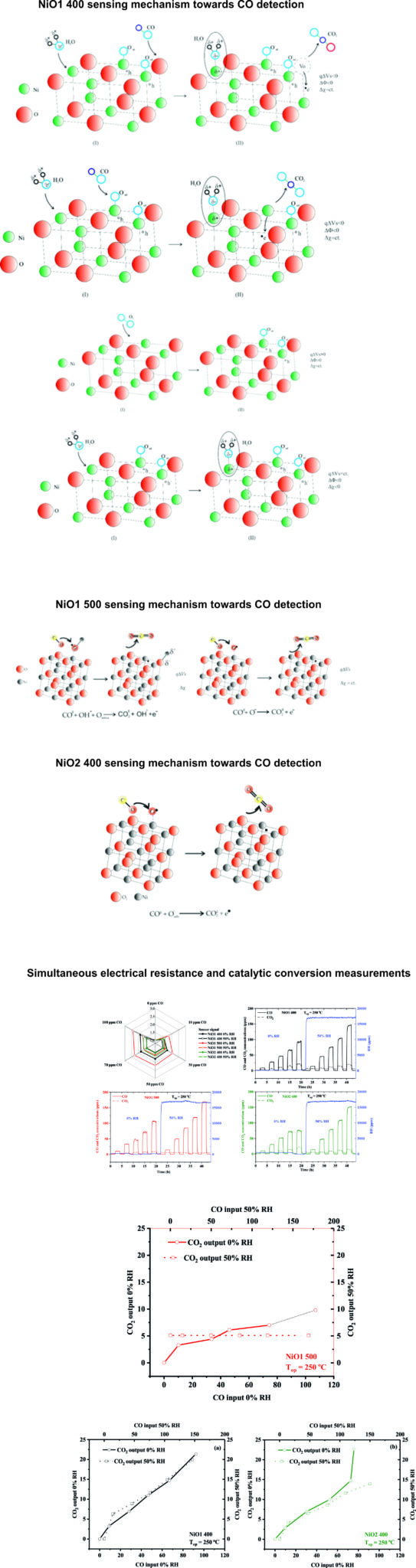
Conclusions regarding the deliverable 3.1.1. By decoupling the interactions of physisorption from those of ionosorption, in the case of the gas-sensitive material NiO1 400, it was observed the possible interaction between CO both with oxygen pre-adsorbed on the surface and with network oxygen. In the case of NiO1 500, carbon monoxide interacts both with the pre-adsorbed oxygen species and with the hydroxyl groups present on the surface. For the NiO2 400 material, CO interaction occurs only with pre-adsorbed oxygen species.
Conclusions regarding the deliverable 3.2.1. With the help of the formalism of quasi-chemical equations, we were able to write the dependence of the coverage ratio with oxygen species on the surface as a function of the bending of the bands as well as of the total free states. In the case of carbon monoxide interacting directly with pre-adsorbed oxygen, we were able to explain the relationship between the coverage ratio, in the Schottky approximation, and the dependence on the average concentration of majority charge carriers on the surface (vacancies).
Conclusions regarding the deliverable 3.2.2. Using the Boltzmann distribution, it was possible to separate the transport phenomena using the theory of diffusion and that of thermoelectronic emission.
From the first theory it follows that the conductance depends both on the surface band bending phenomena and on the intrinsic volume properties. In the case of thermoelectronic emission theory, the conductance depends only on the concentration of charge carriers at the surface. 8. The estimated impact of the results obtained, emphasizing the most significant result obtained. The funded project "Perspectives on detection mechanisms with nickel oxide-based gas sensors" is a direct consequence of experimental studies on metal oxide semiconductors (MOX) with p-type semiconductor character (e.g. NiO). The studied materials in which the effects of exposure to reducing gases (e.g. CO or relative humidity) indicated very pronounced charge transfer processes on the extraction work (ΔΦ) which, however, are not reflected in the changes in electrical resistance (R) of the sensitive gas layer. The explanation lies in the fact that the adsorption of oxygen species on the surface causes the appearance of an accumulation layer of voids, and if we consider the equivalent circuit of the NiO layer as a parallel circuit of resistances corresponding to the accumulation layer on the surface of crystallites (small in value) and of volume (large), we can see that the resistances that change are those of the surface layer and thus the impact in the total resistance of the MOX sensitive layer will be reduced. It is expected that the obtained results will have a great impact in the understanding of different conduction mechanisms, with direct implications in the development of technologies based on field effects (e.g. MOSFET-type gas sensors), where the field modification is controlled by the composition of the surrounding atmosphere.
The stage of achieving the 2021 achievement indicators - two papers published in Chemosensors-MDPI, an ISI rated journal; an Editorial published in Materials-MDPI; a participation in the "Contemporary Solutions for Advanced Materials with high Impact on Society" Workshop, October 11-15, 2021, Bucharest; web page
The stage of achieving the 2022 achievement indicators - two papers published in Chemosensors-MDPI – IF = 4.229, on the subject of the project; a paper published in Sensors and Actuators B. Chemical – IF = 9.221 and one in Materials Letters – IF = 3.574 with thanks to the current project; a participation in the International Conference on Semiconductors (CAS) with an oral presentation during October 12-14, 2022. IEEE Xplore Proceedings 2022 pp. 109-112, https://doi.org/10.1109/CAS56377.2022.9934703; web page.
The stage of achieving the achievement indicators 2023 - a paper published in Sensors and Actuators B. Chemical, a paper under review in Applied Surface Science Ms. Ref. No.: APSUSC-D-23-15783 Title: Atomic-level insight into the structural properties of nanoparticulate NiO for CO sensing on the subject of the project; a paper accepted in the journal Physica E: Low-dimensional Systems and Nanostructures with thanks to the current project; an invited presentation at the International Conference on Semiconductors (CAS) October 11-13, 2023 IEEE Xplore Proceedings 2023 pp. 11-18, https://doi.org/10.1109/CAS59036.2023.10303715; an oral presentation at the International Conference on Semiconductors (CAS) October 11-13, 2023 IEEE Xplore Proceedings 2023 pp. 63-66, https://doi.org/10.1109/CAS59036.2023.10303672 web page.
Articles in ISI-quoted journals
1. A. Stanoiu, A.C. Kuncser, D. Ghica, O.G. Florea, S. Somacescu, C.E. Simion, Sensing Properties of NiO Loaded SnO2 Nanoparticles - Specific Selectivity to H2S, Chemosensors 2021, 9, 125, https://doi.org/10.3390/chemosensors9060125.
2. C.E. Simion, C. Ghica, C.G. Mihalcea, D. Ghica, I. Mercioniu, S. Somacescu, O.G. Florea, A. Stanoiu, Insights about CO Gas-Sensing Mechanism with NiO-Based Gas Sensors-The Influence of Humidity, Chemosensors 2021, 9, 244, https://doi.org/10.3390/chemosensors9090244.
3. C.E. Simion, Special Issue "Advanced Materials for Gas Sensors" Editorial, Materials 2021, 14, 6765, https://doi.org/10.3390/ma14226765.
4. A. Stanoiu, C. Ghica, C.G. Mihalcea, D. Ghica, S. Somacescu, O.G. Florea, C.E. Simion, Effects of Calcination Temperature on CO-Sensing Mechanism for NiO-Based Gas Sensors, Chemosensors 2022, 10, 191, https://doi.org/10.3390/chemosensors10050191.
5. C. Ghica, C.G. Mihalcea, C.E. Simion, I.D. Vlaicu, D. Ghica, I.V. Dinu, O.G. Florea, A. Stanoiu, Influence of relative humidity on CO2 interaction mechanism for Gd-doped SnO2 with respect to pure SnO2 and Gd2O3, Sens. Actuators B. Chem. 2022, 368, 132130, https://doi.org/10.1016/j.snb.2022.132130.
6. Andrei C. Kuncser, Ioana D. Vlaicu, Ion V. Dinu, Cristian E. Simion, Alexandra C. Iacoban, Ovidiu G. Florea, Adelina Stanoiu, The impact of the synthesis temperature on SnO2 morphology and sensitivity to CO2 under in-field conditions, Materials Letters, 2022, 325, 132855, https://doi.org/10.1016/j.matlet.2022.132855.
7. C.E. Simion, O.G. Florea, I. Mercioniu, I.V. Dinu, A. Stanoiu, Gas sensing mechanism involved in NH3 detection with NiO material, International Semiconductor Conference (CAS) 45th Edition, 12-14 October 2022, pg. 109-112 (prezentare orala), Proceedings of the IEEE Xplore https://doi.org/10.1109/CAS56377.2022.9934703
8. A. Stanoiu, C. Ghica, C.G. Mihalcea, D. Ghica, C.E. Simion, The Role of the Synthesis Routes on the CO-Sensing Mechanism of NiO-Based Gas Sensors, Chemosensors 2022, 10, 466, https://doi.org/10.3390/chemosensors10110466.
9. Cristian E. Simion, Ioana D. Vlaicu, Alexandra C. Iacoban, Catalina G. Mihalcea, Corneliu Ghica and Adelina Stanoiu*, The influence of the synthesis method on Gd2O3 morpho-structural properties and sensitivity to CO2 under in-field conditions, Materials Chemistry and Physics 296, 15 February 2023, 127354, https://doi.org/10.1016/j.matchemphys.2023.127354
10. Cristian.E. Simion, Benjamin Junker, Udo Weimar, Adelina Stanoiu, Nicolae Bârsan, Sensing mechanisms of CO and H2 with NiO material – DRIFTS investigations, Sens. Actuators B. Chem. 390, 1 September 2023, 134028, https://doi.org/10.1016/j.snb.2023.134028
11. A. Stanoiu, D. Ghica, C.G. Mihalcea, I.D. Vlaicu, O.G. Florea, S. Bulat, C. Ghica, C.E. Simion*, GHGs detection by tuning the operating temperature of Sn1-xGdxO(4-x)/2, International Semiconductor Conference (CAS), Sinaia, Romania, November 2023, pp. 63-66, (prezentare orala/articol), Proceedings of the IEEE Xplore https://doi.org/10.1109/CAS59036.2023.10303672
12. C. E. Simion, I. V. Dinu, O. G. Florea and A. Stanoiu*, The role of interdigital electrodes on sensing performances with p-type NiO-based gas sensors -link to experiments, International Semiconductor Conference (CAS), Sinaia, Romania, November 2023, pp. 11-18, (prezentare invitata/articol), Proceedings of the IEEE Xplore https://doi.org/10.1109/CAS59036.2023.10303715
13. Ion V. Dinu, Cristian E. Simion, Nicoleta G. Apostol, Ovidiu G. Florea, Catalina G. Mihalcea and Adelina Stanoiu, Conduction mechanism of Gd2O3 induced by CO2 under in-field conditions, accepted in 24 noiembrie 2023 la Physica E: Low-dimensional Systems and Nanostructures, Reference: PHYSE_115862.
14. C. G. Mihalcea, M. Stefan, C. Ghica, O. G. Florea, A. Stanoiu, C. E. Simion, S. Somacescu, D. Ghica, In-depth insight into the structural properties of nanoparticulate NiO for CO sensing, Applied Surface Science 651 (2024) 159252, https://doi.org/10.1016/j.apsusc.2023.159252.
Conference presentations
1. A. Sobetkii, R.E. Irimescu, A.E. Slobozeanu, C.F. Ciobota, U. Cindemir, L. Osterlund, A. Stanoiu, C.E. Simion, R.M. Piticescu, Deposition and characterization of thin films based on nanostructured NiO as sensorial element for detection gases, Workshop on "Contemporary Solutions for Advanced Catalytic Materials with a High Impact on Society", 11-15 October 2021, Bucharest, Romania, (oral presentation) https://chimie.unibuc.ro/edu/greencam/index.php/workshop-2021.
2. C.E. Simion, O.G. Florea, I. Mercioniu, I.V. Dinu, A. Stanoiu, Gas sensing mechanism involved in NH3 detection with NiO material, International Semiconductor Conference (CAS) 45th Edition, 12-14 October 2022, pg. 109-112, oral presentation.
3. A. Stanoiu, D. Ghica, C.G. Mihalcea, I.D. Vlaicu, O.G. Florea, S. Bulat, C. Ghica, C.E. Simion*, GHGs detection by tuning the operating temperature of Sn1-xGdxO(4-x)/2, International Semiconductor Conference (CAS), Sinaia, Romania, November 2023, pp. 63-66, oral presentation.
4. C. E. Simion, I. V. Dinu, O. G. Florea and A. Stanoiu*, The role of interdigital electrodes on sensing performances with p-type NiO-based gas sensors -link to experiments, International Semiconductor Conference (CAS), Sinaia, Romania, November 2023, pp. 11-18, invited talk.
| Nr. Crt. | Name and surname | ID Brainmap | |
| 1 | GHICA Corneliu | cghica@infim.ro | U-1700-029U-5827 |
| 2 | STANOIU Adelina | adelina.stanoiu@infim.ro | U-1700-038M-8092 |
| 3 | GHICA Daniela | ghica@infim.ro | U-1700-035Y-0481 |
| 4 | SIMION Cristian Eugen | simion@infim.ro | U-1700-030F-9765 |
| 5 | KUNCSER Andrei Cristian | andrei.kuncser@infim.ro | U-1700-032N-6988 |
| 6 | MERCIONIU Ionel Florinel | imercioniu@infim.ro | U-1700-039E-8823 |
| 7 | ISTRATE Marian Cosmin | cosmin.istrate@infim.ro | U-1900-061U-8873 |
| 8 | MIHALCEA Catalina | catalina.mihalcea@infim.ro | U-1900-062F-0766 |
| 9 | FLOREA Ovidiu | ovidiu.florea@infim.ro | U-1700-032D-4435 |
| 10 | DINU Ion Viorel | vdinu@infim.ro | U-1700-030A-3912 |
| 11 | SOMACESCU Simona | somacescu.simona@gmail.com | U-1700-039T-4737 |
PROJECTS/ NATIONAL PROJECTS
Copyright © 2026 National Institute of Materials Physics. All Rights Reserved